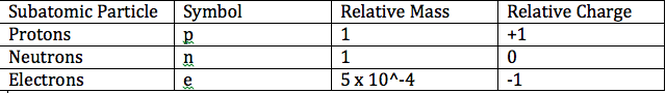
The Structure of an Atom
Atoms are made out of 3 subatomic particles which are the protons, electrons and neutrons. Each atom consists of 2 areas, the nucleus and the electron shells. The nucleus is at the center of the atom and it consists of protons and neutrons. The electrons occupy the empty space around the nucleus and spins in orbits. Each shell can hold a specific number of electrons.
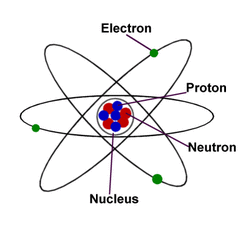
The proton is a positively charged atom that is situated at the center of an atom (the nucleus) and is placed with the neutrons. The neutron don't have any charge. The mass and the radioactivity of the atom is affected by the neutrons. The electron is a negatively charged particle that is found spinning around the outside of the nucleus. The atom is said to have a neutral charge when the number of protons and electrons are the same. The protons in the nucleus attract the electrons since one is positive and the other is negative. Electrons are way more smaller than neutrons and protons. They are about 1800 times smaller.
Despite the fact the electrons are the smallest and have the smallest mass of the three subatomic particles, they control the chemical properties. When an element has different number of electrons, it would be a different element which would have different chemical properties since the electrons control the chemical properties. The different number of electrons will have different arrangements in their electron shells.
Despite the fact the electrons are the smallest and have the smallest mass of the three subatomic particles, they control the chemical properties. When an element has different number of electrons, it would be a different element which would have different chemical properties since the electrons control the chemical properties. The different number of electrons will have different arrangements in their electron shells.