Ions, Isotopes and Radioisotopes
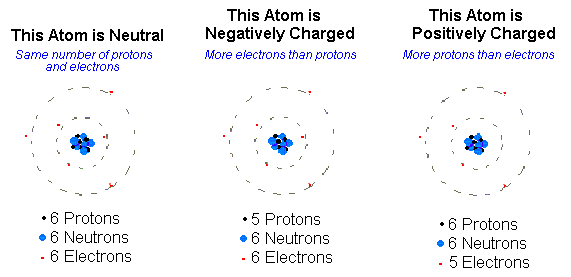
A normal atom has an equal number of positive and negative particles which makes it neutral. That would mean that an atom with a neutral charge is one where the number of protons is equal to the number of electrons. Ions are atoms which have extra or missing electrons. When you are missing an electron or more, you have a positively charged atom, but when you have an extra electron or more you'd have a negatively charged atom. Atoms are not able to gain or lose protons because they are tightly placed in the nucleus, unlike the electrons that lie on the outer region of an atom.
An atom that is missing or has extra neutrons are called Isotopes. Isotopes are the same elements that have two or more forms that contain the same number of protons, however they have a different number of neutrons. Each isotope has different relative atomic masses, so they differ in physical properties but not chemical properties.
Isotopes of the same element always have the same atomic number and element symbol. Isotopes of the same element have the same chemical properties but different physical properties. For example, a lighter isotope will diffuse faster than a heavy one.
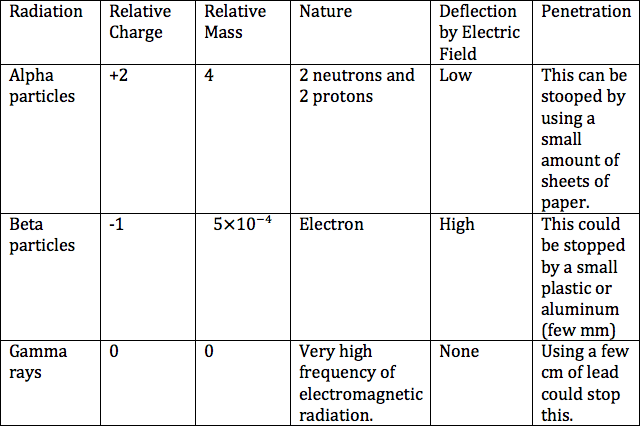
Various chemical elements have unstable nuclides.The nuclei of these chemical elements separate suddenly with the outflow of ionizing radiation. These unstable nuclides are known to be radioactive, therefore they were named radioisotopes. There are 3 types of radiation and could be both found and measured using a tool called Geiger-Muller tube.
An atom that is missing or has extra neutrons are called Isotopes. Isotopes are the same elements that have two or more forms that contain the same number of protons, however they have a different number of neutrons. Each isotope has different relative atomic masses, so they differ in physical properties but not chemical properties.
Isotopes of the same element always have the same atomic number and element symbol. Isotopes of the same element have the same chemical properties but different physical properties. For example, a lighter isotope will diffuse faster than a heavy one.
Various chemical elements have unstable nuclides.The nuclei of these chemical elements separate suddenly with the outflow of ionizing radiation. These unstable nuclides are known to be radioactive, therefore they were named radioisotopes. There are 3 types of radiation and could be both found and measured using a tool called Geiger-Muller tube.
Isotopes could either be stable or unstable. An Isotope will decay over time and will turn into anther element or isotope when it's unstable. Isotopes that are unstable are considered radioactive. Many elements that could be found in nature are made of stable isotopes. Tin is the element with the most stable isotopes with 10 different stable isotopes.
Below is a demonstration of how to calculate the relative atomic mass (this is a screenshot from bbc.co.uk):
Below is a demonstration of how to calculate the relative atomic mass (this is a screenshot from bbc.co.uk):
Different radioisotopes have different uses. Radioisotopes of elements like Cobalt-60, Iodine-131 and Iodine-125 are used in radiotherapy to damage the DNA of cancer cells. This will help prevent them from experiencing cell division.
Americum-241 is an element that has a number of different uses, but most importantly it helps us determine where oil wells should be drilled.
Californium-252 is an element that is used for security reasons in places like airports because they are used for inspecting hidden explosives in the luggage. It is also used for measuring the moist of the materials that could be found in the soil.
Radioisotopes are also used as a method of treating food in order to have a longer shelf life and safer to eat. This procedure is not very different from other treatments such as, canning, freezing and drying.
Americum-241 is an element that has a number of different uses, but most importantly it helps us determine where oil wells should be drilled.
Californium-252 is an element that is used for security reasons in places like airports because they are used for inspecting hidden explosives in the luggage. It is also used for measuring the moist of the materials that could be found in the soil.
Radioisotopes are also used as a method of treating food in order to have a longer shelf life and safer to eat. This procedure is not very different from other treatments such as, canning, freezing and drying.