Ionisation Energy
The first ionisation energy is the minimum energy required to remove the most loose held electron from one mole of isolated gaseous atoms to form 1 mole of gaseous positive ions with a charge of +1.
Ionisation energy is the measure of the amount of energy used to remove an electron from an atom. Since the electrons in the shells are negatively charged and the protons in the nucleus are positively charged, there will be some kind of attraction between them.
There are number of different factors that affect ionisation and those are:
1. Atomic size: When having a small atom (an atom with a small number of energy levels), the electrons stay close to the nucleus and they have a strong nuclear attraction. This means that more ionisation energy is needed to remove electrons from small atoms. On the other hand, in atoms with a larger radius (a big sized atom) the valence electron is away from the nucleus, so they require less nuclear attraction to remove an electron from the atom. In conclusion, it requires more ionisation energy to remove electrons from small atoms than from removing electrons from a big sized atom.
" Therefore, Ionization Energy is directly proportional to Nuclear charge." (Sciencechamps)
2. Nuclear charge: The electrons have more nuclear attraction when increasing the nuclear charge (by adding more protons to the nucleus). So more ionisation energy is needed.
" Therefore, Ionization Energy is directly proportional to Nuclear charge." (Sciencechamps)
3. Shielding effect: The valence electrons are being charged attracted by the positively charged nucleus but are also being repelled by the other electrons from the other shells. The electron-electron repulsion is the shielding effect which decreases the nuclear attraction.
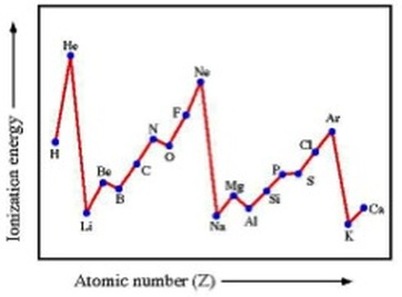
The picture below shows the trends in ionisation energy across the first 20 elements in the periodic table:
Ionisation energy is the measure of the amount of energy used to remove an electron from an atom. Since the electrons in the shells are negatively charged and the protons in the nucleus are positively charged, there will be some kind of attraction between them.
There are number of different factors that affect ionisation and those are:
1. Atomic size: When having a small atom (an atom with a small number of energy levels), the electrons stay close to the nucleus and they have a strong nuclear attraction. This means that more ionisation energy is needed to remove electrons from small atoms. On the other hand, in atoms with a larger radius (a big sized atom) the valence electron is away from the nucleus, so they require less nuclear attraction to remove an electron from the atom. In conclusion, it requires more ionisation energy to remove electrons from small atoms than from removing electrons from a big sized atom.
" Therefore, Ionization Energy is directly proportional to Nuclear charge." (Sciencechamps)
2. Nuclear charge: The electrons have more nuclear attraction when increasing the nuclear charge (by adding more protons to the nucleus). So more ionisation energy is needed.
" Therefore, Ionization Energy is directly proportional to Nuclear charge." (Sciencechamps)
3. Shielding effect: The valence electrons are being charged attracted by the positively charged nucleus but are also being repelled by the other electrons from the other shells. The electron-electron repulsion is the shielding effect which decreases the nuclear attraction.
The picture below shows the trends in ionisation energy across the first 20 elements in the periodic table:
We can see in the picture above that the energy level has a pattern of slowly rising and then suddenly dropping. This shows that every big drop is a change of energy level, but the zig zag's correspond to the change of sub level in an energy level.
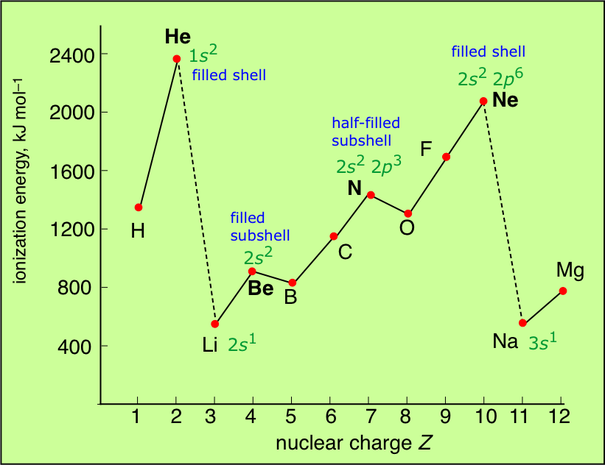
The following picture shows how this graph can tell you how each sub level and energy level is filled up:
The following picture shows how this graph can tell you how each sub level and energy level is filled up:
The picture above is a great evidence to show that energy level and sub levels exist. H and He are elements that have 1 or 2 electrons and that's why they are in the first energy level since the first energy level hold up to 2 electrons. Then the drop shows that we have moved to the 2nd energy level and that's where 8 electrons could be held. The 2nd energy level has 2 sub levels, the first one can hold 2 electrons and the second one can hold 6 electrons. So Li and Be are elements with 3 and 4 electrons, so they fill up the 1st sub level of the 2nd energy level. Then from B to Ne are elements that have 5-10 electrons so we can fill them up in the 2nd sub level.
Hydrogen's electronic structure is 1s1. The only electron is very close to the nucleus and therefore strongly attracted. The ionisation energy is high (1310 kJ mol-1). since there are no electrons screening that one electron from the nucleus.
Helium has a structure of 1s2. It's in the same energy level as hydrogen, since the first energy level can hold up to 2 electrons, so it's also close to the nucleus and unscreened. The only difference is that there are 2 protons attracting the electron instead of one, so the value of the inosiation energy would become much higher and would become (2370 kJ mol-1).
Lithium's structure is 1s2,2s1. Its valence electron is in the second energy level, which is a bit far from the nucleus. A person might argue that it will need more inonisation energy that Helium because it has 3 protons in the nucleus, but that is false because the electron doesn't feel the full pull of the nucleus since it's being screened by 1s2. Lithium's ionisation energy is 519 kJ mol-1.
Hydrogen's electronic structure is 1s1. The only electron is very close to the nucleus and therefore strongly attracted. The ionisation energy is high (1310 kJ mol-1). since there are no electrons screening that one electron from the nucleus.
Helium has a structure of 1s2. It's in the same energy level as hydrogen, since the first energy level can hold up to 2 electrons, so it's also close to the nucleus and unscreened. The only difference is that there are 2 protons attracting the electron instead of one, so the value of the inosiation energy would become much higher and would become (2370 kJ mol-1).
Lithium's structure is 1s2,2s1. Its valence electron is in the second energy level, which is a bit far from the nucleus. A person might argue that it will need more inonisation energy that Helium because it has 3 protons in the nucleus, but that is false because the electron doesn't feel the full pull of the nucleus since it's being screened by 1s2. Lithium's ionisation energy is 519 kJ mol-1.