Atomic Emission Spectrum and Spectroscopy
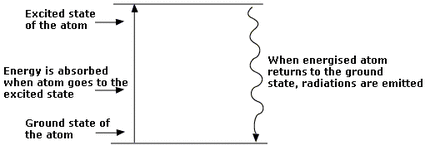
By giving the atom additional energy (such as heat or electricity), the atoms would get excited. When that happens, the electrons would change from the lowest energy level (which is called ground state) to a higher energy level (which is called the excited state). Once the electrons get excited, they will eventually lose their energy, so when that happens, the atom re-emits a photon in the form of light (electromagnetic radiation). One packet of quantum (energy) or photon is released for each electron transition. The energy difference between the excited state and the ground state is equal to the energy of photon. The light that is produced by the excited electrons is consisted of different frequencies and lengths. These are then represented in an atomic emission spectrum that has spectral lines along an increasing frequency scale.
The energy of the photon that is released can be found by using this equation:
E= hf
In the equation above, 'E' represents the energy of a photon (Joules), h represents Planck’s constant (6.63 x 10^-34 J) and the f represents the frequency of light (hertz).
An example of this is:
Calculate the energy of the photon when it has a frequency of 5.2 × 10^14 Hz .
f=5.2 × 10^14
h=6.63 x 10^-34
E= 5.2 × 10^14 x 6.63 x 10^-34
E= 3.4476 × 10^-19 J
So the energy of the photon is 3.45 × 10^-19 J
Atomic emission spectroscopy is a way of chemical analysis that uses the power of light emitted from a fire, arc, or a spark at a wavelength to determine how much of an element is in a specific sample. The instrument that's used to separate the different components of light, depending on their wavelengths is called a spectroscope or a a spectrometer. The wavelengths are then converted to frequencies by using this equation:
F= c/λ
In here c is the speed of light.
The energy of the photon that is released can be found by using this equation:
E= hf
In the equation above, 'E' represents the energy of a photon (Joules), h represents Planck’s constant (6.63 x 10^-34 J) and the f represents the frequency of light (hertz).
An example of this is:
Calculate the energy of the photon when it has a frequency of 5.2 × 10^14 Hz .
f=5.2 × 10^14
h=6.63 x 10^-34
E= 5.2 × 10^14 x 6.63 x 10^-34
E= 3.4476 × 10^-19 J
So the energy of the photon is 3.45 × 10^-19 J
Atomic emission spectroscopy is a way of chemical analysis that uses the power of light emitted from a fire, arc, or a spark at a wavelength to determine how much of an element is in a specific sample. The instrument that's used to separate the different components of light, depending on their wavelengths is called a spectroscope or a a spectrometer. The wavelengths are then converted to frequencies by using this equation:
F= c/λ
In here c is the speed of light.
