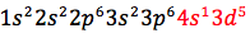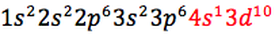Full Electron Configuration of Atoms
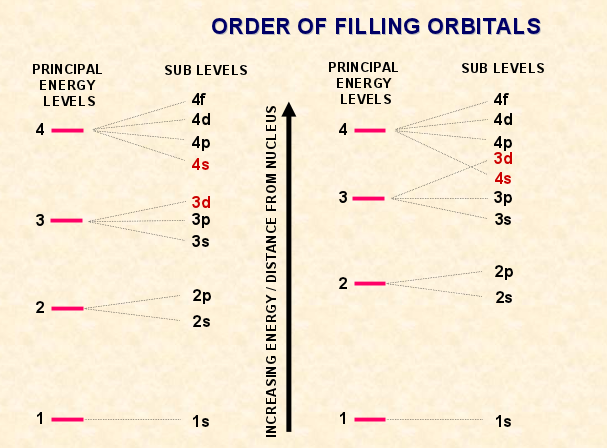
Electrons are arranged according in energy levels. The energy level closest to the nucleus is the most stable energy level and the one with the lowest energy. The electron configuration of an atom helps us know the number of electrons in each sub level in each energy level. The first energy level can only hold 2 electrons, every other energy level can hold up to 8 electrons. The AUFBAU Principle says "electrons enter the lowest energy level", so each electron is assigned to the lowest energy sub level available. The first energy level is the closest one to the nucleus, once it's filled we add electrons one by one until the number of electrons is equal to the number of protons in the nucleus. The outer energy level is called the valence shell since it's the shell that holds the electron that affects the chemical reactions and the bonding. An example of this would be Potassium which has an electronic configuration of 2,8,8,1. The valence electron could be easily lost which makes it unstable, has a high reactivity and a valency of 1. The first sub level that would be filled is the first sub level which is '1s', after that would come the '2s' sub level, then 2p,3s,3p,4s,3d etc.
The following picture shows the order of filling orbitals:
The following picture shows the order of filling orbitals:
Another way to remember this is by knowing the following picture:
Hydrogen is an element which has one proton and one electron. This one electron is assigned to the 1s sub-level which is in the lowest energy sub level in the lowest energy level. Therefore, the electron configuration of hydrogen is written like this:
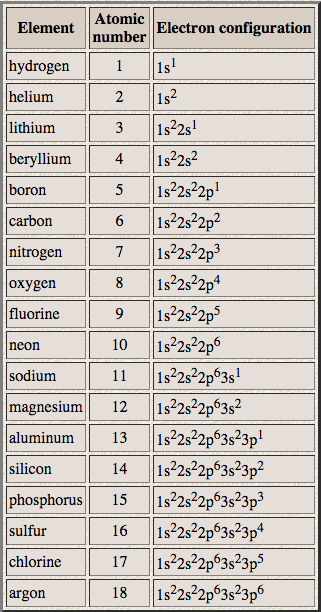
This table shows the electronic configuration of the first 18 elements in the periodic table:
The electron configuration of an atom is how the electrons are arranged when distributed among the sub shells. The periodic table is a tool which is very helpful to determine the electron configuration, but also we have to keep in mind that there are principles and rules such as Heisenberg's uncertainty principle, The Aufbau principle and Pauli's exclusion principle, to look for and follow when putting electrons to different energy level and sub levels.
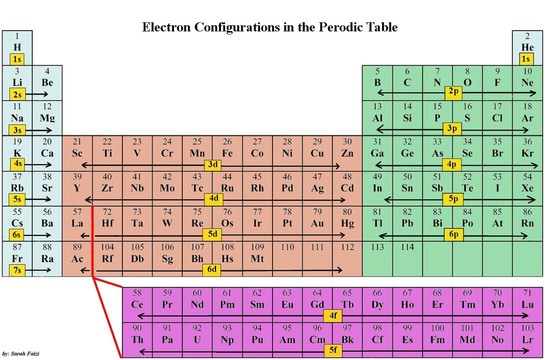
The periodic table helps us in writing the electronic configuration. The pattern in the periodic table helps you identify which element is in which sub level. The following picture will demonstrate what I mean:
The periodic table helps us in writing the electronic configuration. The pattern in the periodic table helps you identify which element is in which sub level. The following picture will demonstrate what I mean:
There are 2 exceptions in the periodic table and those Chromium and Copper, their electronic configuration is as follow:
Chromium (Cr) has an atomic number of 24 and this is how its electronic configuration is written:
Chromium (Cr) has an atomic number of 24 and this is how its electronic configuration is written:
"To achieve a more stable arrangement of lower energy, one of the 4s electrons is promoted into the 3rd to give six unpaired electrons with lower repulsion" (Mr Engstrom's presentation).
Copper (Cu) has an atomic number of 29 and this is how its electronic configuration is written:
Copper (Cu) has an atomic number of 29 and this is how its electronic configuration is written:
"To achieve a more stable arrangement of lower energy, one of the 4s electrons is promoted into the 3d" (Mr Engstrom's Presentation).
As can be seen in the 2 elements above, the 3d sub level is filled before the 4s is filled. This is because the electrons fill the energy sub levels to minimize the energy of the atom.
As can be seen in the 2 elements above, the 3d sub level is filled before the 4s is filled. This is because the electrons fill the energy sub levels to minimize the energy of the atom.