Orbitals and Energy Levels
Electrons are arranged in energy levels around the nucleus of an atom. Each energy level consists of a number of sub levels which are labelled as s,p,d or f. The number of the energy level is equal to the number of sub levels it has. For example, energy level 1 has 1 sub level, energy level 2 has 2 sub levels, energy level 3 has 3 sub levels and so on.
The following table shows the number of sub levels and sub shell of each energy level form 1-4:
The following table shows the number of sub levels and sub shell of each energy level form 1-4:
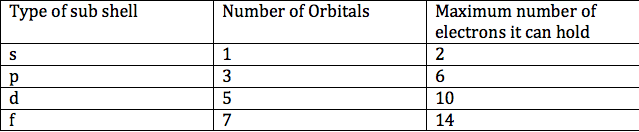
The table below summarizes the number of orbitals and the maximum number of electrons each type of sub shell can hold:
Electrons do not have a specific position in an atom, they do not spin in orbits like we were used to be told in grade 10 and below. Electrons could be anywhere around the atom, but it's very likely to be next to the nucleus if it was in the first energy and very unlikely to be next to the nucleus if it was in the 4th energy level. The four different types of orbitals are s, p, d and f, and each one has a different shape. It's more likely to diffuse when the orbital is larger since the density of the electron is less. The most important shells are the ones that are the furthest from the nucleus because they are involved in the chemical bond.
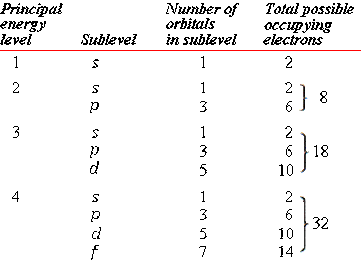
The following table will show the sub shell, number of orbital in each sub shell and how many electrons can be held in each sub shell in each energy level:
The following table will show the sub shell, number of orbital in each sub shell and how many electrons can be held in each sub shell in each energy level:
There are certain principles that help us when arranging electrons in atomic orbitals and they are:
1. Heisenberg's Uncertainty Principle states that you cannot know where an electron's exact place is. We were always taught that they spin in orbits, but that's not the case. We cannot know both the momentum and where it is at the same time, we can only know one at a time.
2. The Aufbau Principle: Electrons fill the lowest available energy level (For example, we fill up 2s before 3s)
3. Pauli's Exclusion Principle: Only two electrons can be filled in each orbital, both in opposite sides because they repel each other.
4. Hund's Rule of Maximum Multiplicity: In a sub shell, electron repel each other and so they enter two different orbitals in the same energy level.
1. Heisenberg's Uncertainty Principle states that you cannot know where an electron's exact place is. We were always taught that they spin in orbits, but that's not the case. We cannot know both the momentum and where it is at the same time, we can only know one at a time.
2. The Aufbau Principle: Electrons fill the lowest available energy level (For example, we fill up 2s before 3s)
3. Pauli's Exclusion Principle: Only two electrons can be filled in each orbital, both in opposite sides because they repel each other.
4. Hund's Rule of Maximum Multiplicity: In a sub shell, electron repel each other and so they enter two different orbitals in the same energy level.